"Water, water, every where Nor any drop to drink."
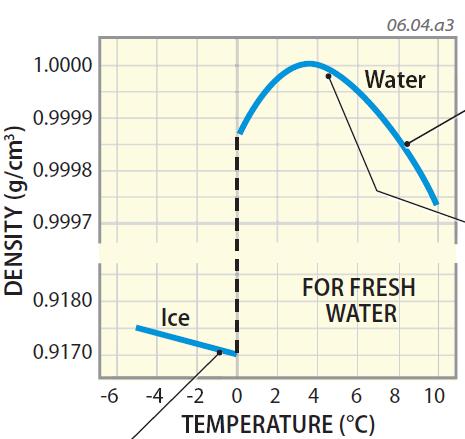
This is described famously by a line from Samuel Taylor Coleridge's The Rime of the Ancient Mariner: The factual accuracy of this article is disputed.Įven on a ship or island in the middle of the ocean, there can be a "shortage of water" meaning a shortage of fresh water. One theory is that plate tectonics result in salt being forced under the continental land masses, where it is again slowly leached to the surface. Ocean salinity has been stable for billions of years, most likely as a consequence of a chemical/tectonic system which removes as much salt as is deposited for instance, sodium and chloride sinks include evaporite deposits, pore water burial, and reactions with seafloor basalts Since the ocean's creation, sodium is no longer leached out of the ocean floor, but instead is captured in sedimentary layers covering the bed of the ocean. The sodium and chloride ions subsequently became the most abundant constituents of sea salt. The presence of the other dominant ion of salt, chloride, results from "outgassing" of chloride (as hydrochloric acid) with other gases from Earth's interior via volcanos and hydrothermal vents. In addition, sodium was leached out of the ocean floor when the oceans first formed. Halley termed this process "continental weathering". Halley noted that of the small number of lakes in the world without ocean outlets (such as the Dead Sea and the Caspian Sea, see endorheic basin), most have high salt content. Upon reaching the ocean, these salts would be retained and concentrated as the process of evaporation (see Hydrologic cycle) removed the water. Scientific theories behind the origins of sea salt started with Sir Edmond Halley in 1715, who proposed that salt and other minerals were carried into the sea by rivers, having been leached out of the ground by rainfall runoff. Total Molal Composition of Seawater (Salinity = 35) Component Differences like these are due to the varying residence times of seawater solutes sodium and chlorine have very long residence times, while calcium (vital for carbonate formation) tends to precipitate out much more quickly. For instance, although seawater is ~2.8 times more enriched with bicarbonate than river water based on molarity, the percentage of bicarbonate in seawater as a ratio of all dissolved ions is far lower than in river water bicarbonate ions constitute 48% of river water solutes, but only 0.41% of all seawater ions. However, the ratios of various solutes differ dramatically. Seawater is more enriched in dissolved ions of all types than fresh water. s -1, and varies with water temperature and pressure.Ĭompositional differences from fresh water.The speed of sound in seawater is about 1500 m Seawater pH is limited to the range 7.5 to 8.4. Deep in the ocean, under high pressure, seawater can reach a density of 1050 kg m -3, depending on the temperature and salinity.The density of surface seawater ranges from about 1020 to 1029 kg The salinity in isolated seas and salt-water lakes (for example, the Dead Sea) can be considerably greater.
The most saline open sea is the Red Sea, where high rates of evaporation, low precipitation and river inflow, and confined circulation result in the formation of unusually salty seawater.

Where mixing occurs with fresh water runoff from river mouths or near melting glaciers, seawater can be substantially less saline. Every mole of water has a mass of \[18.015$ molecules.Main article: Salinity Seawater composition (by mass)Īlthough the vast majority of seawater has a salinity of between 3.1% and 3.8%, seawater is not uniformly saline throughout the world. Given: a density of 1.00 g/ml shows that every milliliter of water has a mass of 1.00 g.ġ8 ml has a mass of $18ml \times 1g/1ml = 18g$ Density is represented as the ratio of mass per unit of volume. We use the density of water at $4^\circ $C and the volume of the sample to find its mass by using the molar mass of water. When the temperature decreases to 0 degrees C, density starts to decrease and it gets near to the freezing point of water. The density of water is directly proportional to salinity and indirectly proportional to temperature.Īt 4 degrees C, the density of water increases. The density is also known as ‘specific gravity’ when an object is made up of molecules. Hint:Density is equal to the ratio of the mass per unit volume of a substance but the mass is the same as the weight on earth.
